Currently, there are no approved treatments for Fragile X Syndrome. The BLOOM clinical trial is studying if MRM-3379 can safely and effectively help improve certain symptoms in male participants with Fragile X Syndrome.
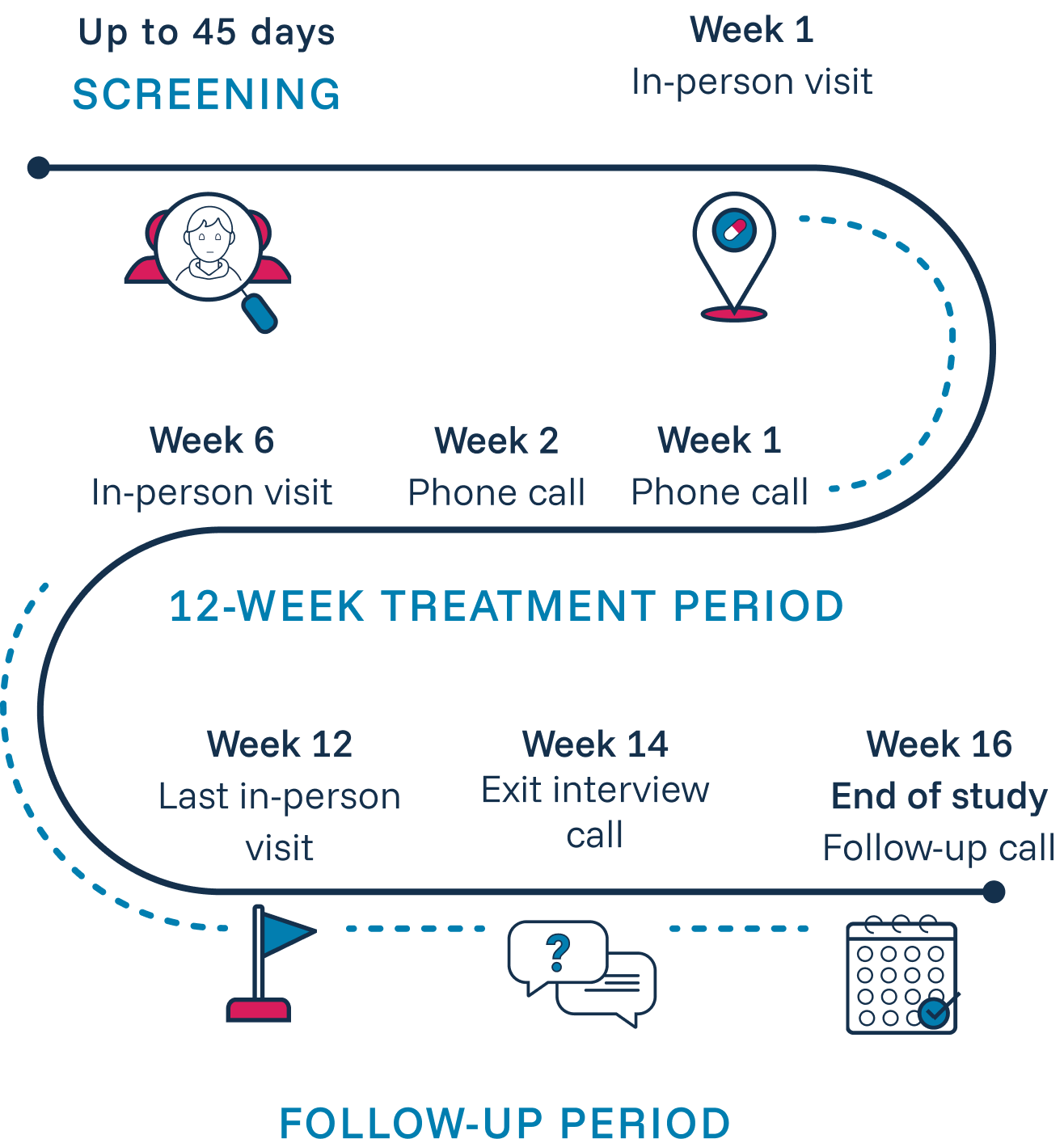
BLOOM is a multicenter, double-blind, randomized, placebo-controlled trial enrolling male patients aged 16–45. In parallel, male patients aged 13–15 will be enrolled in an open-label arm to receive MRM-3379 twice daily for 12 weeks.1

![]()

Cognition, measured as change from baseline at Week 12 on the NIH-TCB Crystallized Cognition Composite (Picture Vocabulary Test (PVT) and Oral Reading Recognition Test (ORRT))
Review the following criteria to determine if your male patients with Fragile X Syndrome qualify for the BLOOM clinical trial.
 Review the study information at clinicaltrials.gov
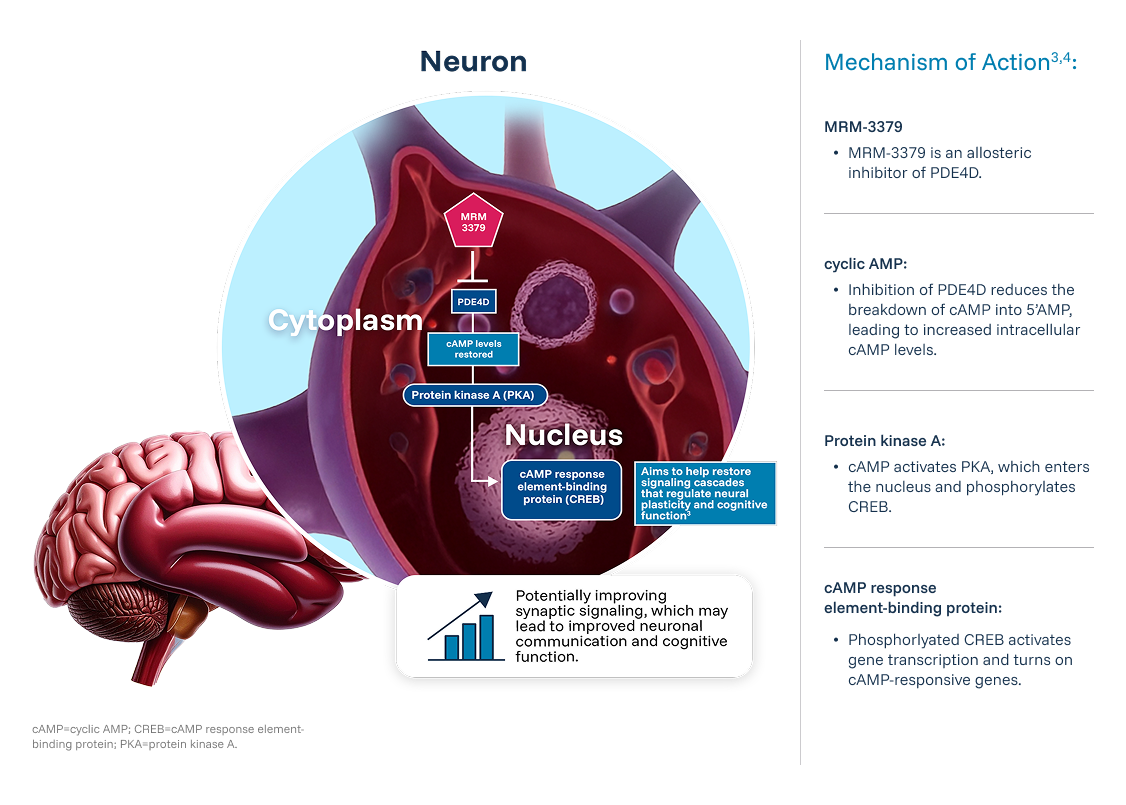
Review the study information at clinicaltrials.gov In Fragile X Syndrome, loss of Fragile X messenger ribonucleoprotein (FMRP) is correlated with increasing disease severity. Previous studies have shown that cyclic adenosine monophosphate (cAMP) level was increased when FMRP was overexpressed and decreased when FMRP was absent. Increasing cAMP levels may help correct the signaling disruption caused by Fragile X Syndrome.3,4
MRM-3379 is an investigational medication designed to block phosphodiesterase‑4D (PDE4D) in the brain to increase cAMP levels.3,4